
Spesolimab prevents generalized pustular psoriasis flares in EFFISAYIL™ 2 trial
Data demonstrate superiority vs. placebo in preventing generalized pustular psoriasis (GPP) flares up to 48 weeks[1]
Prevention of unpredictable flares addresses a high unmet patient need[2,3]
Results build on the EFFISAYIL™ 1 trial, which demonstrated rapid and sustained pustular and skin clearance in flaring GPP patients treated with spesolimab for 12 weeks[2]
-

About Generalized Pustular Psoriasis (Graphic: Business Wire)
-
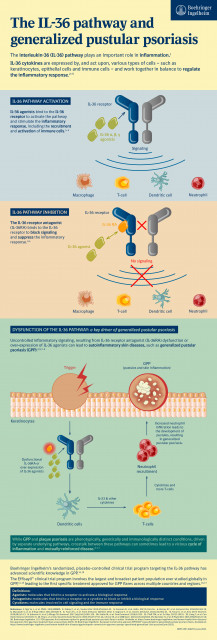
The IL-36 pathway and generalized pustular psoriasis (Graphic: Business Wire)
INGELHEIM, GERMANY--(Business Wire / Korea Newswire)--Today, at the 25th World Congress of Dermatology (WCD), Boehringer Ingelheim presented new late-breaking data from the EFFISAYIL™ 2 trial showing that spesolimab significantly reduced the risk of GPP flares by 84% over 48 weeks compared to placebo. Furthermore, the trial with 123 patients demonstrated no flares after Week 4 of spesolimab treatment in the high dose group.[1]
“EFFISAYIL™ 2 is the first and largest multinational randomized clinical trial to evaluate a treatment for the prevention of GPP flares,” said Bruce Strober, MD, PhD, Clinical Professor, Dermatology, Yale University and Central Connecticut Dermatology. “These results provide further compelling clinical evidence for the role IL-36 signaling plays in the pathogenesis of GPP. Moving forward, our hope is that dermatologists not only have a specific treatment for GPP flares, but that we can effectively prevent them in the future.”
GPP flares, characterized by painful pustules all over the body, often require emergency care and can lead to serious, life-threatening complications such as sepsis, shock and multisystem organ failure.[3] In addition to the acute distress of a GPP flare, living with the uncertainty of the impact of the next flare can have a significant emotional burden on patients.[4]
“At IFPA we meet people living with GPP, who share their story of physical pain due to the symptoms of the disease, and the mental burden that comes with the flares,” said Frida Dunger Johnsson, Executive Director, IFPA. “Any progress made to ease the burden of the disease improves the quality of life of people living with GPP.”
“Through our comprehensive EFFISAYIL™ clinical program we have already delivered spesolimab as a major advancement for flaring GPP patients,” said Carinne Brouillon, Head of Human Pharma, Boehringer Ingelheim. “The EFFISAYIL™ 2 trial results build on this success bringing us closer to achieving our ultimate goal of a flare-free future for everyone living with GPP.”
Spesolimab (marketed as SPEVIGO®) has recently received Breakthrough Therapy Designation (BTD) from the U.S. Food and Drug Administration (FDA) and the Center for Drug Evaluation (CDE) of China’s National Medical Products Administration (NMPA) as an investigational treatment for the prevention of GPP flares.
In the EFFISAYIL™ 2 trial, spesolimab was shown to have a favorable safety profile, with a similar incidence of patients with adverse events across spesolimab and placebo treatment arms.[1]
About spesolimab
Spesolimab is a novel, humanized, selective antibody that specifically blocks the activation of the interleukin-36 receptor (IL-36R), a signaling pathway within the immune system shown to be involved in the pathogenesis of several autoinflammatory diseases, including GPP.[2,5,6]
It is the first approved treatment for GPP flares in adults that has been evaluated in a statistically-powered, randomized, placebo-controlled trial.[2] To date, SPEVIGO® (spesolimab) has been approved by regulatory authorities in almost 40 countries including the US, Japan, Mainland China, and the European Union to treat GPP flares in adults.[7,8]
Spesolimab is also under investigation for the treatment of other IL-36 mediated skin diseases.[9]
For the full press release including ‘Notes to Editors’ and references please visit: press release. https://bit.ly/44rEQzq
View source version on businesswire.com: https://www.businesswire.com/news/home/20230703881533/en/
Website: https://www.boehringer-ingelheim.com
Contact
Boehringer Ingelheim
Corporate Communications
Media + PR
Laura Lessenich
+49 (6132) 77-173436
This is a news release distributed by Korea Newswire on behalf of this company.

