
Microba Commences Phase I Clinical Trial for IBD Therapeutic
First participants have been successfully dosed in a Phase I Clinical Trial of MAP 315, Microba’s lead Therapeutic Program in Inflammatory Bowel Disease.
MAP 315, is a novel live biotherapeutic product being developed for the treatment of ulcerative colitis and was discovered and developed using Microba’s data-driven Therapeutics Platform.
This milestone transitions Microba into a clinical stage drug development company with results from the trial expected in December 2023.
A video presentation of this milestone by Microba CEO Dr Luke Reid and SVP, Therapeutics Prof Trent Munro can be viewed here https://ir.microba.com/presentations/
-
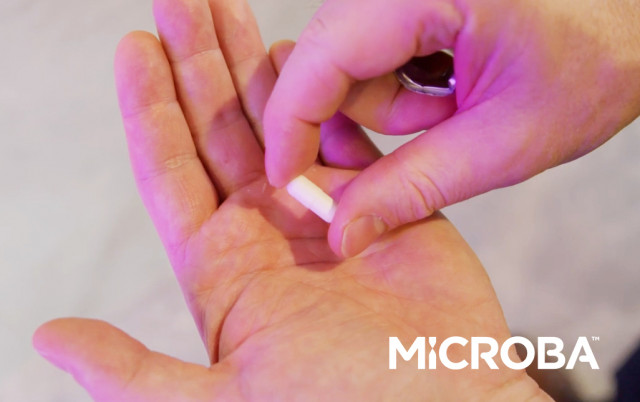
MAP 315, is a novel live biotherapeutic product being developed for the treatment of ulcerative colitis and was discovered and developed using Microba’s data-driven Therapeutics Platform. (Photo: Business Wire)
BRISBANE, AUSTRALIA--(Business Wire / Korea Newswire)--Microba Life Sciences Limited (ASX: MAP) (“Microba” or the “Company”) today announces that the first participants have been successfully dosed in the Phase I Clinical Trial of MAP 315, in Microba’s Therapeutic Program in Inflammatory Bowel Disease (IBD). This milestone brings Microba’s drug candidate MAP 315 into clinical development as a potential new treatment option for the millions of people suffering from ulcerative colitis.
Professor Trent Munro, SVP of Therapeutics at Microba said: “Microba has now matured into a clinical stage drug development company. I am proud of our team and what we have built to bring novel microbiome drugs all the way from human-data guided discovery, through to an orally delivered drug product now dosed in a first in human clinical trial. We are uniquely positioned at the forefront of drug development from the microbiome and excited about the future potential impact of MAP 315 as a new treatment for Inflammatory Bowel Disease sufferers.”
The Phase I clinical trial is a randomised, double-blind, placebo-controlled study to evaluate the safety, tolerability and pharmacokinetics of MAP 315 in 32 healthy adults. The trial is being conducted by Nucleus Network in Melbourne, Australia, under the Company’s Human Research Ethics Committee (HREC) approval and in alignment with formal feedback received from an FDA pre-IND engagement.
MAP 315 Phase 1 Clinical Trial Details
The clinical trial is titled “A Phase 1, Randomised, Double-Blind, Placebo-Controlled, Study to Evaluate the Safety, Tolerability and Pharmacokinetics of MAP 315 in Healthy Adults”. The study will enroll 2 cohorts of 16 participants each, who will be randomised 3:1 to receive MAP 315 or its matching placebo for 14 consecutive days (2 weeks). Results from the trial are expected to be available in December 2023. Full study details can also be found in the Australian New Zealand Clinical Trials Registry (ANZCTR) under study ID: ACTRN12623000291684.
Microba’s Novel Drug Candidate MAP 315
MAP 315 was identified using Microba’s unique analysis of its large proprietary human databank, demonstrating that this previously uncharacterised and novel bacterial species is commonly observed in healthy individuals but consistently deficient in individuals with IBD. Subsequent pre-clinical investigation of MAP 315 through both in vitro and in vivo models demonstrated that MAP 315 promotes epithelial restitution and mucosal healing - biological activities that are critical for sustained disease remission but not adequately addressed through existing therapy.
MAP 315 is a live biotherapeutic product (LBP) consisting of lyophilised bacteria in an enteric coated capsule which is orally administered and being developed as an investigational product for the treatment of ulcerative colitis (UC). Microba worked in partnership with leading microbiome contract drug manufacturer Bacthera to produce GMP compliant MAP 315 at large-scale in their state-of-the-art facilities in Europe to supply material for the MAP 315 clinical trial.
Inflammatory Bowel Disease - Large unmet need & commercial opportunity
IBD causes prolonged inflammation of the digestive tract and now affects more than 7 million people globally, with this number increasing each year[1]. Ulcerative colitis (UC) is one of the two major forms of IBD, which results in inflammation and ulcers (sores) in the digestive tract, causing a debilitating chronic condition. Patients are currently treated with anti-inflammatory and immunomodulatory medication to dampen the disease and control symptoms, often with significant side effects. These available treatment options commonly fail, with more than 50% of patients unable to achieve sustained remission[2], which sees them experiencing regular episodes of inflammation, diarrhoea, bleeding and abdominal pain[3], with as many as 25% of patients requiring hospitalisation[4]. The market for UC treatment was valued at US$7.5 billion in 2020 and is forecast to grow to US$10.8 billion by 2030[5]. Microba’s novel drug candidate MAP315 presents an opportunity to improve the current standard of care and treatment for millions of people suffering from the debilitating effects of UC.
Therapeutic Platform & Programs
There is a growing body of evidence that the gut microbiome plays a central role in the maintenance of health and the development of chronic disease. With microbiome-based therapeutics now in clinical development and the first FDA approvals, these novel drugs represent an exciting new opportunity for the treatment of chronic diseases that are underserved by current pharmaceuticals.
Microba is at the forefront of this field using its advanced proprietary metagenomics technology developed by leading Australian researchers in the top 1% of cited researchers globally. Using this technology, Microba has established a data-driven platform for drug discovery and development from the human gut microbiome. This platform leverages a large, growing, proprietary databank collected through the Company’s Microbiome Testing Services, and is generating multiple potent therapeutic candidates to address chronic diseases. Microba has established three therapeutic programs spanning IBD, Immuno-Oncology and Autoimmune Diseases, with lead candidate MAP 315 under the Company’s IBD program the first program to enter human clinical trials.
About Microba Life Sciences Limited
Microba Life Sciences is a precision microbiome company driven to improve human health. With world-leading technology for measuring the human gut microbiome, Microba is driving the discovery and development of novel therapeutics for major chronic diseases and delivering gut microbiome testing services globally to researchers, clinicians, and consumers. Through partnerships with leading organisations, Microba is powering the discovery of new relationships between the microbiome, health and disease for the development of new health solutions.
[1] https://www.thelancet.com/journals/langas/article/PIIS2468-1253(19)30333-4/fulltext
[2] https://www.crohnscolitisfoundation.org/sites/default/files/2019-02/Updated%20IBD%20Factbook.pdf
[3] Scribano, M.L. Adverse events of IBD therapies. Inflamm Bowel Dis. (2008). https://doi.org/10.1002/ibd.20702.
[4] Pola, S. et al. Strategies for the care of adults hospitalized for active ulcerative colitis. Clin Gastroenterol Hepatol. (2012). https://doi.org/10.1016/j.cgh.2012.07.006.
[5] https://www.nature.com/articles/d41573-021-00194-5, https://www.alliedmarketresearch.com/ulcerative-colitis-market
View source version on businesswire.com: https://www.businesswire.com/news/home/20230627143323/en/
Website: https://microba.com/
View Korean version of this release
Contact
Microba Life Sciences Limited
This is a news release distributed by Korea Newswire on behalf of this company.

